Nizampura, Vadodara, Gujarat
- GST NO. : 24AAHFB6183E1ZM
+91-7490991897, +91-9824037528, +91-9328211833
| Business Type | Manufacturer, Exporter, Supplier, Retailer |
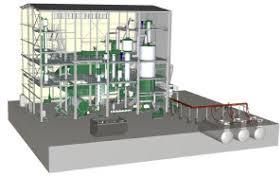
| Type | CNA Plant |
| Weight | As Per Requirement |
| Voltage | As Per Requirement |
| Click to view more | |
Product Details
Nitric Acid Concentration
Concentrated Nitric Acid CNA ( i.e. high-strength nitric acid at 98 -99% w/w.) is an important chemical for Chemical & Pharmaceutical industries in general (like polymers & plastics, dyestuff, bulk drugs, explosives, Fertilizers and other important organic nitro-compounds). This acid is frequently used as "nitrating acid".
Industrially manufactured Nitric acid is obtained as a weak acid solution, at a concentration of about 65% -67% w/w (max.), which is of no use whatsoever for the various processes stated above,
Owing to the physical behaviour of the nrtric acid / water system, it is not possible to remove excess water from this weak acid by a fractional distillation process because this solution forms an azeotrope, a constant acid / water boiling composition at about 68% w/w. acid.
To concentrate this acid further an extractive distillation process by adding concentrated sulphuric acid needs to be employed. While the water goes this sulphuric acid gets diluted, which can be again re-concentrated and reused for the concentration of dilute nitric acid again.
Reconcentration of weak Sulphuric Acid
The dilute sulphuric acid stream coming off the bottom of the Extractive Distillation Column is reconcentrated back to the same service by using BVR's sulphuric acid re-concentration plant, which can be coupled directly with the nitric acid concentration system so that no cooling of the weak sulphuric acid is necessary, thus saving a large amount of heating energy for the economical benefit of the user.
NOx Absorption
The NOx gases escaping from the Nitric acid Concentration System, along with bleaching air, are absored in BVR's special NOx absorption plant and is recycled back into the main system as an aqueous solution of nitric acid, at concentration comparable to those of the nrtric acid feed to the same System : the residual gases vented to the atmosphere have NOx. content below the acceptable values as per environmental norms.
Pre-concentration of weak Nitric Acid solution
Whenever a solution of Nitric Acid is too weak for direct Extractive Distillation (typically below 45% -50% w/w.), it can be pre-concentrated to its azeotropic grade by our vacuum Fractional Distillation plant.
Looking for "Concentrated Nitric Acid CNA Plant" ?
Explore More Products