Nizampura, Vadodara, Gujarat
- GST NO. : 24AAHFB6183E1ZM
+91-7490991897, +91-9824037528, +91-9328211833
| Business Type | Manufacturer, Exporter, Supplier |
| Type | As Per Requirement |
| Application | As Per Requirement |
| Grade | As Per Requirement |
| Click to view more | |
Product Details
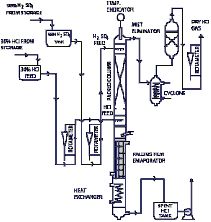
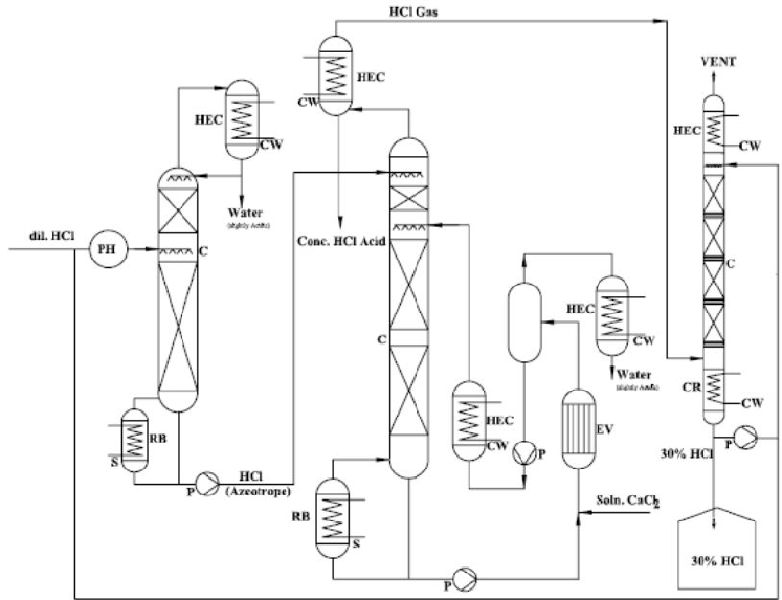
The material system hydrogen chloride/water displays a maximum azeotrope at a boiling temperature of 108.6°C,for a system pressure of 1 bar and a HCl concentration of 20.2wt%. If the acid concentration is lower than the azeotrope mixture, the acid can be concentrated only upto the azeotropic point. Further concentration needs special procedures. We have considered the most economical contemporary technology-“Extractive Rectification & Absorption Technology”.
Extractive Rectification & Absorption Technology
As mentioned this is an economical technology and is presented in illustration below the azeotrope point is suppressed as best as possible and increases the relative volatility of HCl. The relative volatility may be impacted by change of the activity coefficients. This is achieved by addition of a third ingredient, the extractive agent, which displays strong hygroscopic properties. Conc. HCL & HCl gas is produced at the head of the extractive rectification column upon choice of an appropriate extraction agent. In the column the extraction agent is diluted with water, which is extracted in a selective fashion from the inflowing mixture. Subsequently, the extractive agent is recycled and newly fed into the extractive rectification process.
Sulphuric acid, and aqueous solutions of CaCl2 are appropriate extraction agents for the concentration of hydrochloric acid.
- Sulphuric Acid Route
- CaCl2 Route

Looking for "HCl Concentration Plant" ?
Explore More Products